Balancing pool and spa water is perhaps the fundamental job of the service pro, and total alkalinity is a key parameter in that process.
What is Total Alkalinity?Total alkalinity is the amount of bicarbonate, plus the much smaller amount of carbonate, plus (in stabilized pools) the amount of cyanurate ions. It is a measure of the pH buffering capacity of water; that is, the ability of water to resist a pH change. Alkalinity is generally expressed in terms of the equivalent concentration of calcium carbonate in ppm. |
The proper total alkalinity level in pool or spa water provides buffering so pH does not swing in and out of the proper range in response to sanitizer addition, bather load or other factors. With too little alkalinity, there will not be enough buffering and the pH may quickly drift out of the proper range. At excessively high bicarbonate/carbonate alkalinity, there will be a tendency for the pH of the water to drift upward, due to the rapid escape of carbon dioxide from the water into the air.
RELATED: Using Air and Acid to Quickly Get Perfect pH/Alkalinity
In addition to these effects on pH, the portion of the alkalinity coming from carbonate and bicarbonate, called carbonate alkalinity, also affects calcium carbonate saturation. The maintenance of calcium carbonate concentration within the recommended range reduces the tendency of pool water to scale or degrade pool surfaces.
When total alkalinity is properly adjusted, optimum buffering results are achieved; pH, swimmer comfort, sanitizer efficacy, water balance and clarity are more easily maintained.
How To Adjust
The Right Amount of TATotal alkalinity should be maintained between a minimum 60 and a maximum of 180 ppm as CaCO3 (calcium carbonate). Ideally, total alkalinity should be maintained between 80 and 100 ppm as CaCO3 where electrolytic chlorine generators, calcium hypochlorite, lithium hypochlorite and sodium hypochlorite are used, because these sanitizers cause the pH to rise. The ideal range where sodium dichlor, trichlor, chlorine gas and bromine compounds are used is between 100 and 120 ppm as CaCO3, because these sanitizers will cause the pH to drift downwards. Certain systems such as PHMB are relatively unaffected by and do not impact total alkalinity. |
Total alkalinity should be corrected before adjusting pH or sanitizer levels. (See notes below regarding "Adjusting for the Effect of Cyanuric Acid on Total Alkalinity.")
To reduce total alkalinity, acid is added to the water. Approximately 2.1 pounds of sodium bisulfate (94 percent) or 1.6 pints of muriatic acid (31 percent) will reduce the total alkalinity of 10,000 gallons of water by 10 ppm.
To increase total alkalinity, use sodium bicarbonate. Approximately 1.5 pounds of sodium bicarbonate (100 percent) will raise the total alkalinity of 10,000 gallons of water by 10 ppm.
Adjusting for Effect of Cyanuric Acid on Total Alkalinity:
Dichlor and trichlor sanitizers release cyanuric acid, which serves to stabilize the chlorine sanitizer. Cyanuric acid stabilizer may be added separately as well. The cyanurate system is a weak buffer and will contribute to the total alkalinity measurement.
RELATED: Test Killers — Common Water Testing Interferences and How to Avoid Them
To determine the carbonate alkalinity in a stabilized pool or spa:
1. Measure the pH
2. Measure total alkalinity (TA)
3. Measure cyanuric acid concentration (CA). If the CA is 90ppm or greater, it may be necessary to dilute the pool water sample with tap water to get an accurate reading.
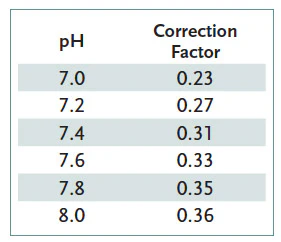
4. Note the Cyanuric Acid Correction Factor in Table 1, based on the pH of the water.
5. Adjust the CA and subtract this result from the total alkalinity for the actual or corrected TA.
6. Formula : Measured TA — (CA x Cyanuric Acid Correction Factor) = Carbonate Alkalinity
Example: pH is 7.4. Total Alkalinity measurement (Measured TA) is 110 ppm. Cyanuric Acid level is 100 ppm.
Cyanuric Acid Correction factor at pH 7.4 is 0.31. (See table above)
Using the formula:
110 ppm — (100 x 0.31) = 110 — 31 = 79 ppm carbonate alkalinity
APSP Recreational Water Quality CommitteeRWQC Chairman James Egan, Ph.D. Gregory Garrett Kenneth Gregory Brett Gereau Chris Kareis, Ph.D. Jody O'Grady David Oxley Shajee Siddiqui Kim Skinner Roy Vore, Ph.D. John Weber Lonza |